PROJECT DETAILS
- Category Early Research
Populations evolve in response to changes in abiotic environments, like climate, as well as to changes in genetic environments, like the internal genetic environment of other genes (epistasis) or the social milieu of parents and other members of the population. In all cases, the rate of adaptation depends upon the frequency of the selective environment.
As part of my doctoral dissertation, I conducted empirical research to understand how phenotypic plasticity contributes to range expansion using a worldwide aquatic invasive snail, Potamopyrgus antipodarum (New Zealand Mud Snails). We have seen a rapid spread from initial points of introduction in the Western United States. Montana State University has an excellent set of resources on the web detailing the spread across the Western United States.
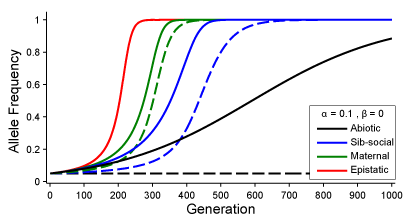
In Drown and Wade (2014), I compare the rate at which individuals adapt to different kinds of environments. In particular, we are contrasting adaptation to an external, non-genetic environment with adaptation to two genetic environments (maternal and epistatic). In all models, the frequency of the selective background, whether external, maternal, or epistatic, determines the strength of selection on a gene, whose direct effect is the environmental adaptation. Adaptation in response to the maternal or the epistatic environment is different from adaptation to an abiotic environment because selection changes the frequency of biotic environments, whereas, the frequency of the external environment does not change as the population adapts to it. Selection creates positive associations between genes of the selective environment and the genes responding to it. This results in a runaway process of coevolution between genes and genetic environments that is much faster than adaptation to the external, non-genetic environment. Furthermore, the coadaptive process is biased toward cooperative genetic interactions and away from genetic conflict: genetic coadaptation is a self-accelerating, runaway process while genetic conflict is a self-braking, diminishing process.
- Dybdahl, M. F., & Drown, D. M. (2011). The absence of genotypic diversity in a successful parthenogenetic invader. Biological Invasions, 13, 1663-1672. https://doi.org/10.1007/s10530-010-9923-4
- Drown, D. M., Levri, E. P., & Dybdahl, M. F. (2011). Invasive genotypes are opportunistic specialists not general purpose genotypes. Evolutionary Applications, 4, 132-143. https://doi.org/10.1111/j.1752-4571.2010.00149.x
- Drown, D. M., & Wade, M. J. (2014). Runaway coevolution: adaptation to heritable and nonheritable environments. Evolution, 68(10), 3039-3046. https://doi.org/10.1111/evo.12470